When a substance changes from one state to another, what happens to the mass of that substance?
7.3: Phase Changes
- Page ID
- 16107
- Determine the heat associated with a phase alter.
Matter tin be in one of several different states, including a gas, liquid, or solid state. The amount of energy in molecules of matter determines the state of thing.
- A gas is a land of matter in which atoms or molecules have enough energy to move freely. The molecules come into contact with ane another only when they randomly collide.
- A liquid is a state of affair in which atoms or molecules are constantly in contact merely accept enough free energy to keep changing positions relative to one some other.
- A solid is a state of matter in which atoms or molecules practise not have plenty energy to move. They are constantly in contact and in fixed positions relative to i another.
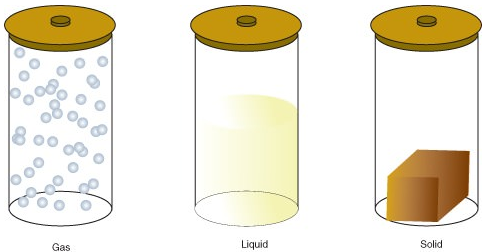
The following are the changes of country:
| Solid → Liquid | Melting or fusion |
| Liquid → Gas | Vaporization |
| Liquid → Solid | Freezing |
| Gas → Liquid | Condensation |
| Solid → Gas | Sublimation |
- If estrus is added to a substance, such as in melting, vaporization, and sublimation, the process is endothermic. In this instance, estrus is increasing the speed of the molecules causing them motion faster (examples: solid to liquid; liquid to gas; solid to gas).
- If estrus is removed from a substance, such as in freezing and condensation, then the process is exothermic. In this example, oestrus is decreasing the speed of the molecules causing them movement slower (examples: liquid to solid; gas to liquid). These changes release estrus to the surround.
- The corporeality of heat needed to modify a sample from solid to liquid would be the aforementioned to contrary from liquid to solid. The only divergence is the direction of heat transfer.
Characterization each of the post-obit processes every bit endothermic or exothermic.
- h2o boiling
- ice forming on a swimming
Solution
- endothermic - yous must put a pan of h2o on the stove and requite it rut in order to go water to boil. Because you are adding rut/free energy, the reaction is endothermic.
- exothermic - think of ice forming in your freezer instead. You put water into the freezer, which takes heat out of the h2o, to get it to freeze. Considering heat is beingness pulled out of the h2o, information technology is exothermic. Rut is leaving.
Exercise \(\PageIndex{i}\)
Label each of the post-obit processes every bit endothermic or exothermic.
- water vapor condensing
- gold melting
- Answer
-
a. exothermic
b. endothermic
A phase change is a physical process in which a substance goes from 1 phase to another. Ordinarily the alter occurs when calculation or removing oestrus at a particular temperature, known as the melting betoken or the boiling point of the substance. The melting point is the temperature at which the substance goes from a solid to a liquid (or from a liquid to a solid). The boiling point is the temperature at which a substance goes from a liquid to a gas (or from a gas to a liquid). The nature of the phase change depends on the direction of the heat transfer. Heat going into a substance changes information technology from a solid to a liquid or a liquid to a gas. Removing heat from a substance changes a gas to a liquid or a liquid to a solid.
Two primal points are worth emphasizing. First, at a substance's melting bespeak or boiling point, 2 phases can exist simultaneously. Have water (HtwoO) as an example. On the Celsius scale, H2O has a melting point of 0°C and a humid point of 100°C. At 0°C, both the solid and liquid phases of H2O can coexist. Still, if rut is added, some of the solid H2O will melt and turn into liquid H2O. If oestrus is removed, the opposite happens: some of the liquid H2O turns into solid H2O. A similar process tin occur at 100°C: calculation rut increases the corporeality of gaseous H2O, while removing heat increases the corporeality of liquid H2O (Figure \(\PageIndex{one}\)).
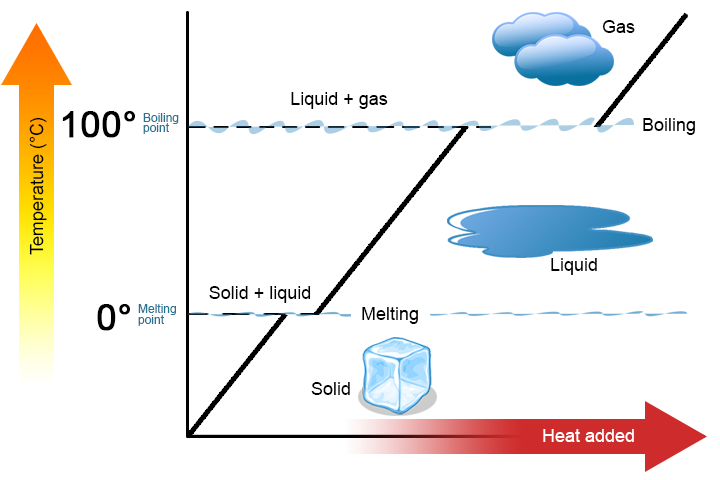
Water is a proficient substance to utilise as an instance because many people are already familiar with it. Other substances take melting points and humid points besides.
Second, every bit shown in Figure \(\PageIndex{1}\), the temperature of a substance does not change as the substance goes from one phase to another. In other words, stage changes are isothermal (isothermal ways "constant temperature"). Again, consider H2O as an instance. Solid water (water ice) can exist at 0°C. If heat is added to water ice at 0°C, some of the solid changes phase to make liquid, which is also at 0°C. Remember, the solid and liquid phases of H2O can coexist at 0°C. Just after all of the solid has melted into liquid does the addition of heat change the temperature of the substance.
For each stage change of a substance, there is a characteristic quantity of heat needed to perform the phase change per gram (or per mole) of textile. The heat of fusion (ΔH fus) is the amount of rut per gram (or per mole) required for a phase change that occurs at the melting point. The oestrus of vaporization (ΔH vap) is the amount of estrus per gram (or per mole) required for a phase modify that occurs at the boiling indicate. If you know the total number of grams or moles of cloth, you lot can use the ΔH fus or the ΔH vap to determine the full heat being transferred for melting or solidification using these expressions:
\[\text{heat} = n \times ΔH_{fus} \label{Eq1a}\]
wher e \(n\) is th e number of moles and \(ΔH_{fus}\) is expressed in free energy/mole or
\[\text{estrus} = m \times ΔH_{fus} \characterization{Eq1b}\]
where \(thousand\) is the mass in grams and \(ΔH_{fus}\) is expressed in energy/gram.
For the boiling or condensation, employ these expressions:
\[\text{estrus} = due north \times ΔH_{vap} \label{Eq2a}\]
wher eastward \(n\) is the number of moles) and \(ΔH_{vap}\) is expressed in free energy/mole or
\[\text{estrus} = one thousand \times ΔH_{vap} \label{Eq2b}\]
wh ere \(m\) i s the mass in grams and \(ΔH_{vap}\) is expressed in energy/gram.
Retrieve that a phase change depends on the management of the oestrus transfer. If heat transfers in, solids become liquids, and liquids get solids at the melting and boiling points, respectively. If heat transfers out, liquids solidify, and gases condense into liquids. At these points, at that place are no changes in temperature every bit reflected in the above equations.
How much heat is necessary to cook 55.viii g of ice (solid HtwoO) at 0°C? The heat of fusion of H2O is 79.nine cal/g.
Solution
Nosotros can utilize the human relationship between heat and the heat of fusion (Equation \(\PageIndex{1}\)) to determine how many cal of heat are needed to cook this ice:
\[ \begin{marshal*} \ce{heat} &= \ce{one thousand \times ΔH_{fus}} \\[4pt] \mathrm{heat} &= \mathrm{(55.8\: \cancel{g})\left(\dfrac{79.9\: cal}{\cancel{g}}\right)=4,460\: cal} \cease{align*}\]
How much heat is necessary to vaporize 685 g of HtwoO at 100°C? The estrus of vaporization of H2O is 540 cal/g.
- Answer
-
\[ \begin{align*} \ce{heat} &= \ce{grand \times ΔH_{vap}} \\[4pt] \mathrm{rut} &= \mathrm{(685\: \cancel{thousand})\left(\dfrac{540\: cal}{\cancel{g}}\right)=370,000\: cal} \end{align*}\]
Table \(\PageIndex{one}\) lists the heats of fusion and vaporization for some common substances. Annotation the units on these quantities; when you use these values in problem solving, make sure that the other variables in your adding are expressed in units consistent with the units in the specific heats or the heats of fusion and vaporization.
| Substance | ΔH fus (cal/g) | ΔH vap (cal/one thousand) |
|---|---|---|
| aluminum (Al) | 94.0 | 2,602 |
| gold (Au) | 15.3 | 409 |
| fe (Fe) | 63.2 | i,504 |
| water (HiiO) | 79.9 | 540 |
| sodium chloride (NaCl) | 123.5 | 691 |
| ethanol (C2HfiveOH) | 45.2 | 200.3 |
| benzene (CviH6) | xxx.4 | 94.1 |
In that location is also a phase modify where a solid goes directly to a gas:
\[\text{solid} \rightarrow \text{gas} \label{Eq3}\]
This phase change is chosen sublimation. Each substance has a characteristic heat of sublimation associated with this process. For case, the oestrus of sublimation (ΔH sub) of HtwoO is 620 cal/m.
We encounter sublimation in several ways. Y'all may already be familiar with dry water ice, which is simply solid carbon dioxide (COii). At −78.5°C (−109°F), solid carbon dioxide sublimes, changing directly from the solid phase to the gas stage:
\[\mathrm{CO_2(due south) \xrightarrow{-78.v^\circ C} CO_2(g)} \label{Eq4}\]
Solid carbon dioxide is called dry water ice because it does non pass through the liquid stage. Instead, it does directly to the gas phase. (Carbon dioxide can exist as liquid but only under high force per unit area.) Dry ice has many applied uses, including the long-term preservation of medical samples.
Even at temperatures below 0°C, solid H2O volition slowly sublime. For example, a thin layer of snow or frost on the footing may slowly disappear as the solid HiiO sublimes, even though the outside temperature may be beneath the freezing point of water. Similarly, ice cubes in a freezer may become smaller over time. Although frozen, the solid water slowly sublimes, redepositing on the colder cooling elements of the freezer, which necessitates periodic defrosting (frost-gratuitous freezers minimize this redeposition). Lowering the temperature in a freezer will reduce the need to defrost as often.
Under similar circumstances, h2o volition also sublime from frozen foods (e.one thousand., meats or vegetables), giving them an unattractive, mottled appearance called freezer burn. It is not really a "burn," and the food has not necessarily gone bad, although it looks unappetizing. Freezer burn down can be minimized by lowering a freezer's temperature and past wrapping foods tightly so water does not have any infinite to sublime into.
Concept Review Exercises
- Explicate what happens when heat flows into or out of a substance at its melting point or boiling point.
- How does the corporeality of oestrus required for a phase change relate to the mass of the substance?
- What is the management of heat transfer in boiling water?
- What is the management of estrus transfer in freezing water?
- What is the direction of heat transfer in sweating?
Answers
one. The energy goes into changing the stage, non the temperature.
ii. The corporeality of heat is a constant per gram of substance.
iii. Boiling. Heat is being added to the water to get it from the liquid state to the gas land.
4. Freezing. Heat is exiting the organization in club to go from liquid to solid. Some other style to look at it is to consider the contrary process of melting. Free energy is consumed (endothermic) to melt ice (solid to liquid) so the opposite process (liquid to solid) must be exothermic.
5. Sweating. Heat is consumed to evaporate the moisture on your skin which lowers your temperature.
Key Takeaway
- In that location is an free energy alter associated with any stage modify.
Exercises
-
How much energy is needed to melt 43.viii one thousand of Au at its melting point of one,064°C?
-
How much energy is given off when 563.8 g of NaCl solidifies at its freezing point of 801°C?
-
What mass of ice tin be melted past 558 cal of energy?
-
How much ethanol (CtwoH5OH) in grams can freeze at its freezing point if 1,225 cal of heat are removed?
-
What is the rut of vaporization of a substance if 10,776 cal are required to vaporize 5.05 g? Express your final reply in joules per gram.
-
If 1,650 cal of oestrus are required to vaporize a sample that has a heat of vaporization of 137 cal/g, what is the mass of the sample?
-
What is the rut of fusion of water in calories per mole?
-
What is the estrus of vaporization of benzene (C6H6) in calories per mole?
-
What is the heat of vaporization of gold in calories per mole?
-
What is the heat of fusion of iron in calories per mole?
Answers
-
670 cal
2. 69,630 cal
-
eight,930 J/g
6. 12.0 g
-
1,440 cal/mol
eight. 7,350 cal/mol
9. 80,600 cal/mol
ten. 3,530 cal/mol
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/The_Basics_of_GOB_Chemistry_%28Ball_et_al.%29/07:_Energy_and_Chemical_Processes/7.03:_Phase_Changes
0 Response to "When a substance changes from one state to another, what happens to the mass of that substance?"
Post a Comment